In today's post we present a new development in the field of renewable energies. In previous posts we have talked about new technologies that help maximise renewable energies, such as solar windows or floating photovoltaic panels, as well as new ways of generating clean energy with plants, such as bio-photovoltaic energy.
These are examples of ways to create green energy from natural resources without generating a negative environmental impact. Nevertheless, if they are to be 100% effective, renewable energies still face a major challenge: storing the energy produced.
The MIT solution
Since these methods depend on meteorological conditions such as the wind or sun, they cannot be relied on to always generate energy, which poses a problem for continuous supply. In order to advance the energy transition, a team of researchers at MIT has come up with a solution to store wind and solar energy.
It involves a rechargeable battery with electrochemical technology that can supply energy for more than 24 hours.
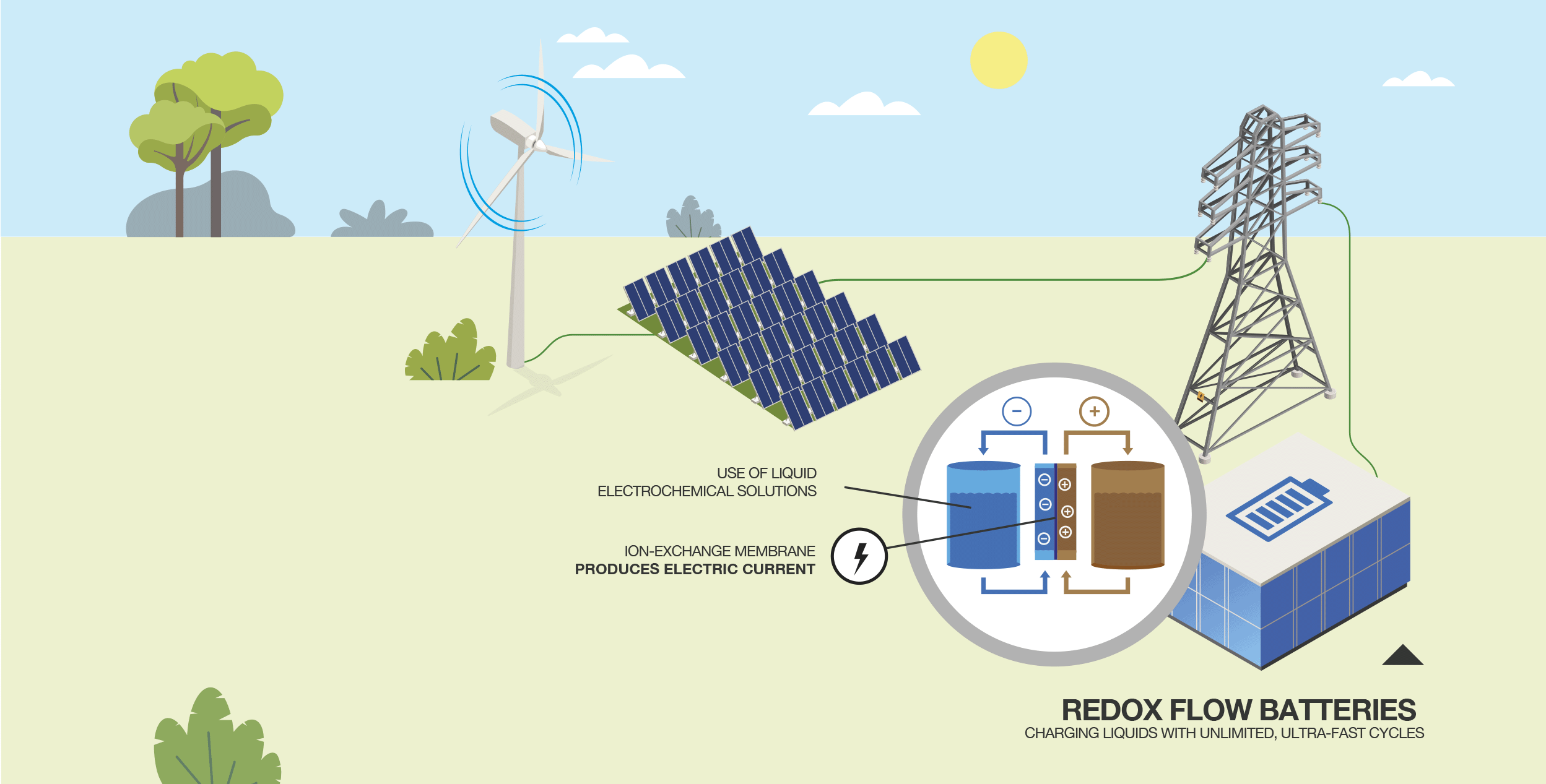
How does a semi-solid flow battery work?
Also known as a rechargeable zinc-manganese dioxide battery, it works by using two chemical components that generate an exchange of positive and negative ions. The ion flows react and convert electrical energy into chemical energy, which charges the battery.
According to the researchers, this method for storing renewable energy is cheaper and more cost-effective than any system currently available. They also point out that it can be expanded on a large scale.
Narayanan, the lead contact on the project, said, “The next step is to take our battery system and build it up. Our research also points the way to other chemistries that could be developed under the semi-solid flow battery platform, so we could be seeing this kind of technology used for energy storage in our lifetimes.”
Source of information:
https://elperiodicodelaenergia.com/el-mit-encuentra-la-solucion-para-el-almacenamiento-de-eolica-y-solar-a-gran-escala-las-baterias-de-flujo-semisolido/
https://www.imnovation-hub.com/es/energia/almacenamiento-energia-renovable/?_adin=02021864894


